Prepared by Mawlana Mateen A. Khan MD, Dr. Ramzan Judge PharmD, Dr. Samad Tirmizi PharmD, and Mufti Adil Farooki MD.
Summary:
Pfizer-BioNtech and Moderna COVID-19 vaccines are ḥalāl and permissible to take.
Summary page for all assessed COVID-19 vaccines
Discussion:
The first class of vaccine to come to market in the United States are mRNA vaccines developed by BioNTech/ Pfizer and Moderna. mRNA vaccines are novel vaccines encapsulating genetic material in lipid nanoparticles. It is thought these particles fuse with the cell membrane of a eukaryote to deliver the mRNA into its cytoplasm. Here, it is translated into the active antigen. Unlike previous vaccines, production of mRNA vaccines do not employ any cell lines or growth medium.[i]

Figure 1: Drawing of a lipid nanoparticle
Pfizer-BioNtech & Moderna COVID Vaccines
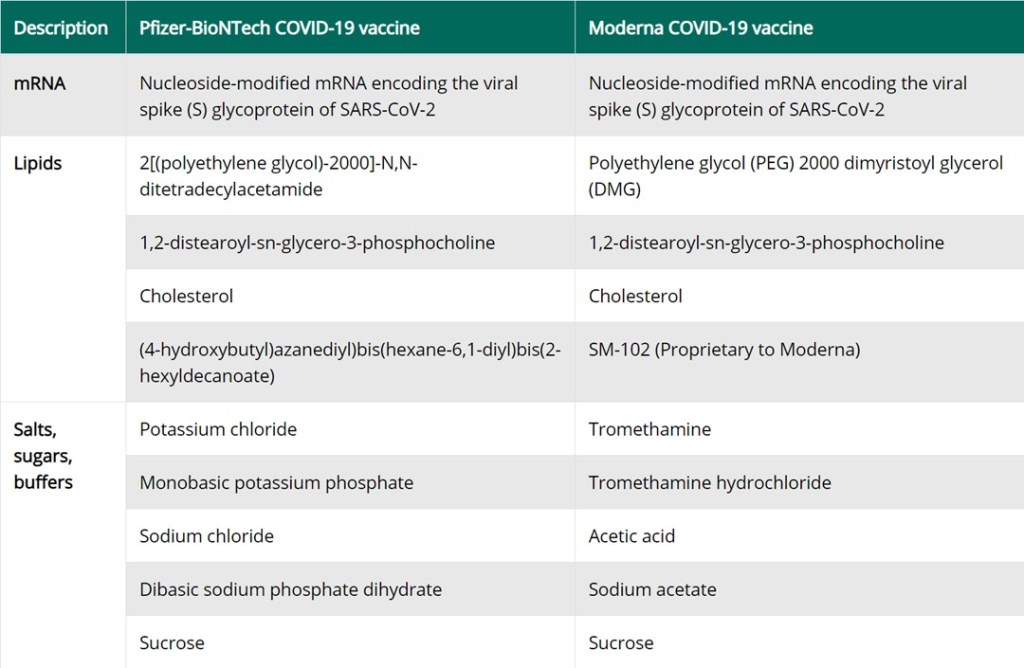
The vaccines developed by Pfizer/ BioNTech and Moderna are mRNA vaccines named BNT162b2 and mRNA-1273 respectively. [ii] The mRNA encodes for the receptor binding domain of the virus’s S1 protein. It is entirely synthetic. There are nine other ingredients (Table 1[iii]). Ingredients 2-5 of each vaccine make up the lipid nanoparticle. Although, these components can be sourced from plant or animal sources, we have assurances from the manufacturers and government agencies that the vaccines do not contain any components of animal origin. [iv] The rest of its components are excipients[v] of ḥalāl origin. Therefore, all ingredients of the Pfizer/BioNTech and Moderna COVID vaccines are ḥalāl, being either non-animal derived synthetic components or excipients.
[i] Annette B. Vogel, Isis Kanevsky et al. A prefusion SARS-CoV-2 spike RNA vaccine is highly immunogenic and prevents lung infection in non-human primates. BioRxiv, September 08, 2020. https://doi.org/10.1101/2020.09.08.280818
Corbett, K.S., Edwards, D.K., Leist, S.R. et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 586, 567–571 (2020). https://doi.org/10.1038/s41586-020-2622-0
[ii] For a better general understanding of Pfizer-BioNTech’s vaccine, see: https://biontech.de/covid-19
[iii] https://www.cdc.gov/vaccines/covid-19/info-by-product/clinical-considerations.html. Accessed December 24, 2020.
[iv] “The Medicines and Healthcare products Agency can confirm that the Pfizer/BioNTech COVID-19 vaccine does not contain any components of animal origin.”
https://www.gov.uk/government/publications/regulatory-approval-of-pfizer-biontech-vaccine-for-covid-19. Accessed 12/5/2020. Confirmation via email correspondence with MHRA on December 17, 2020.
The vaccine provided in the UK is identical in composition to what is provided in the US. (Verbal confirmation from Pfizer representative on December 4, 2020.)
“Our vaccine is free of animal ingredients. The production of it is synthetic.” (Email correspondence from BioNTech. December 7, 2020.)
Moderna vaccine does not contain animal derivatives. (Verbal confirmations from Moderna MSL(s) on December 23 & 24, 2020.)
“Moderna COVID-19 Vaccine does not contain any preservatives, antibiotics, adjuvants, or human- or animal-derived materials.” (Official written statement from Moderna dated December 27, 2020. Also: https://covid-vaccine.canada.ca/info/pdf/moderna-covid-19-vaccine-pm1.pdf. Accessed December 28, 2020).
[v] The excipients as listed in Table 1 include salts, buffers, and sugar. The Pfizer/BioNtech vaccine contains four salts including table salt. Moderna vaccine uses tromethamine, an ingredient found in commonly used medications. These ingredients are primarily used to balance pH. Additionally, Moderna vaccine includes acetic acid, the main component of vinegar, and its salt.


